 
Σ x σ p ≥ ℏ 2 is called the momentum operator in position space. The formal inequality relating the standard deviation of position σ x and the standard deviation of momentum σ p was derived by Earle Hesse Kennard later that year and by Hermann Weyl in 1928: In the published 1927 paper, Heisenberg originally concluded that the uncertainty principle was Δ pΔ q ≈ h using the full Planck constant. For a detailed proof of the Heisenberg inequality that explains every term, you can check GB Folland's Lectures on Partial Differential Equations (pdf. 
2\pi comes from deriving a Fourier transform and the 2 that remains comes from playing around with the terms. Introduced first in 1927 by German physicist Werner Heisenberg, the uncertainty principle states that the more precisely the position of some particle is determined, the less precisely its momentum can be predicted from initial conditions, and vice versa. Actually Heisenberg's inequality naturally comes from using Fourier Analysis. Such paired-variables are known as complementary variables or canonically conjugate variables. In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of mathematical inequalities asserting a fundamental limit to the product of the accuracy of certain related pairs of measurements on a quantum system, such as position, x, and momentum, p. Uncertainty principle of Heisenberg, 1927. In fact, in the abstract, Heisenberg immediately refers to limitations of joint measurements later in the paper, he links this with a statement of the uncertainty relation for the widths of a Gaussian wave function and its Fourier transform. Now, here's the fun part: you can show that the spacial distribution of protons in the nucleus is the Fourier transform of the momentum distribution.Īnd bingo, a measurement of the size of the nucleus.ĭo it with a polarized target and you can get info on the shape as well.Canonical commutation rule for position q and momentum p variables of a particle, 1927. of the uncertainty principle discussed here are already manifest, if only expressed rather vaguely. (This is what nuclear physicists call "quasi-elastic scattering".) Now, if (1) we are shooting a beam of electrons at a stationary target, (2) we have a precision measurement of the momenta of the incident and scattered electrons and the ejected proton, (3) we are willing to neglect excitation energy of the remnant nucleus, and (4) we assume that $p$ mostly did not interact with the remnant after being scattered, we know the momentum of the proton inside the nucleus at the time it was struck.Ĭollect enough statistics on this and we have sampled the proton momentum distribution of the nucleus.  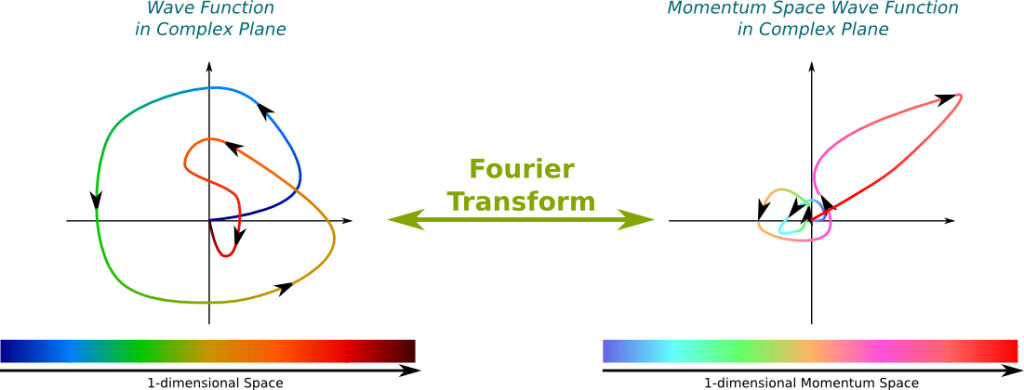
Where $A$ represents that target nucleus and $B$ the remnant after we bounce a proton out. What we do is scatter things off of the component parts of the nucleus. Now, electron microscopy can just about provide vague picture of a medium or large atom as a out-of-focus ball, but there is no hope of employing that technique to something orders of magnitude smaller. What is the shape and size of a atomic nucleus?įrom Rutherford we learned that the nucleus is rather a lot smaller than the atom as a whole. Given that leftaroundabout and vonjd have addressed the fundamental place of the Fourier transform in the formalism, let me talk a little about an experimental application. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
